
Our vision
A reversible and safe therapy for diabetes patients
The historically recent development that the majority of people have sufficient food at their disposal is accompanied by the mass spread of a new group of diseases: metabolic syndrome and obesity in particular.
We would like to offer a reversible therapy for patients who are at an earlier stage of the disease, in which a tube prosthesis is anchored in the stomach and upper small intestine as part of an outpatient gastroscopy.
We want to offer a safe, effective and easy-to-use product that closes the gap between the conservative forms of therapy available to date and irreversible surgery.
Our product
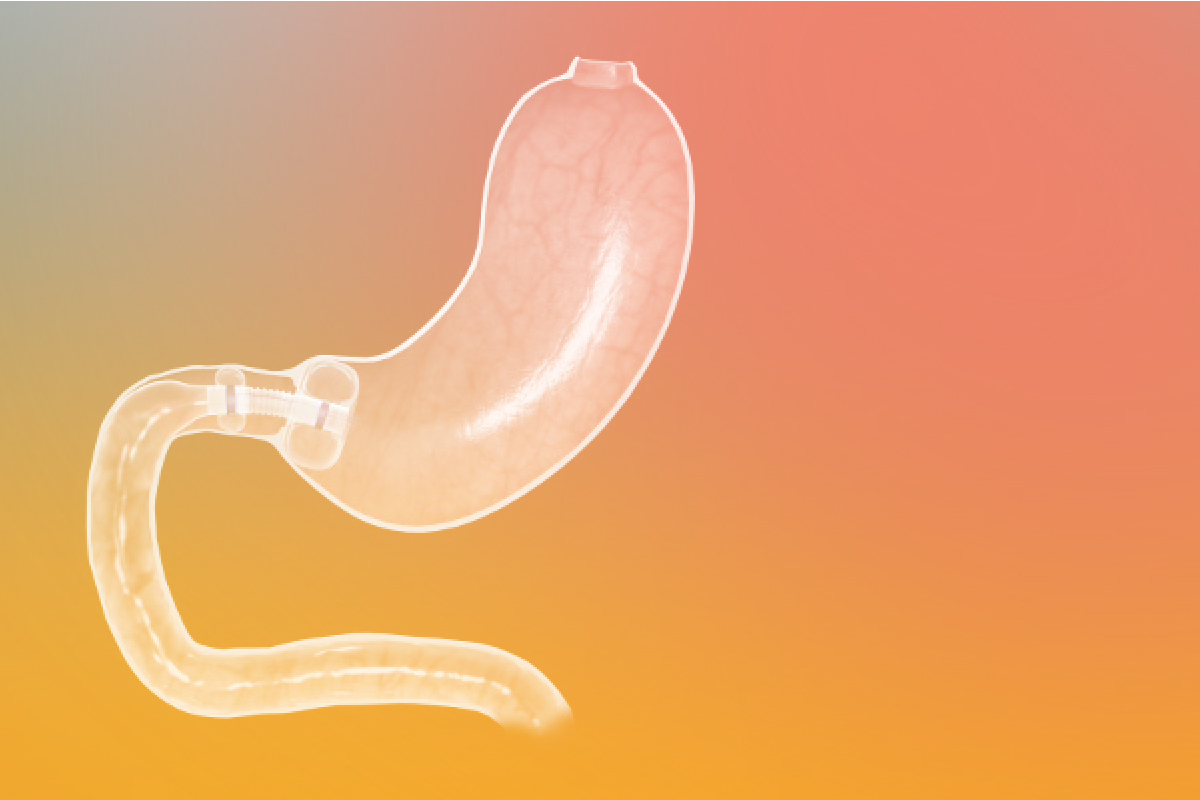
The Trans-Duodenal Barrier
The Trans-Duodenal Barrier is an endoscopically implantable tubular prosthesis made of polyurethane that lines the duodenum and fixes itself to the gastric outlet with two air-filled balloons.
It is inserted during a gastroscopy (gastroscopy) and removed again after 12 months. Similar to bypass surgery, this improves diabetes and reduces weight.
” For users: To the explantation instructions
News
TDC launches pivotal clinical trial for Trans-Duodenal Barrier
Novel endoscopic treatment method for diabetes and obesity from Mannheim being tested nationwide
Trans-Duodenal Concepts GmbH started the clinical approval study for its endoscopic implant Trans-Duodenal Barrier on May 4, 2026 following approval by the BfArM and approval by all responsible ethics committees.

Ovesco takes the next step in metabolic bariatric endoscopy and invests in Transduodenal Concepts
Tübingen and Mannheim, August 26, 2025 – Ovesco Endoscopy AG from Tübingen is investing almost €1 million in Trans-Duodenal Concepts GmbH, Mannheim, to jointly advance innovative solutions in the field of metabolic bariatric endoscopy.
Transduodenal Concepts at the BioRN Annual Conference 2024
This year, Trans-Duodenal Concepts GmbH is participating in the BioRN Annual Conference 2024 (BAC) as an exhibitor. Take advantage of this great opportunity to get to know one of the most vibrant life science ecosystems and learn about our latest technologies on the expo floor.

Robert Schavan joins the Transduodenal Concepts team
Trans-Duodenal Concepts GmbH is proud to announce that we have been able to recruit Mr. Robert Schavan, a proven expert with international experience and a network in the healthcare sector, as Chief Operating Officer from March 2024.

New podcast on the topic of metabolic endoscopy with Prof. Kähler
A podcast on the topic of metabolic endoscopy has been published in the Good Morning, Endoscopy! series, in which Prof. Dr. Alexander Meining, Würzburg, conducts an interview with the CEO of TDC, Prof. Dr. Georg Kähler. The fundamental developments in the endoscopic treatment of metabolic syndrome and the TD Barrier are placed in the context of the current situation.

TD-Barrier awarded 2nd place in the German Medical Award 2023
At Medica Düsseldorf, Prof. Kähler was awarded 2nd prize at the German Medical Award in the presence of NRW Health Minister Karl-Josef Laumann.
Our team
Experts in their field

Prof. Dr. Georg Kähler
Founder & CEO
Dept. Head of endoscopy, Univ. Medicine Mannheim, University of Heidelberg
former President of the german Society for Endoscopy

Robert Schavan
COO
More than 30 years of experience in the medical technology industry in
development, production, approval
QM, sales & marketing

Maher Kasraoui
Dipl.Ing. Plastics Technology
Process Engineer Plastic Bonding Technologies Mechanical Joining Blow Molding
investors
Early stage investment. Opportunity in a rapidly growing market.
There are already 60 million diabetes patients in the EU alone, 26.1 million patients with fatty liver disease and 81.3 million obesity sufferers – and the trend is rising sharply.
With its product, Trans-Duodenal Concepts closes the gap between surgical and conservative therapy for diabetes, the treatment of fatty liver disease and obesity treatment by combining an innovative endoscopic technique with ultra-thin polyurethane balloons.
Following clinical studies, CE certification and multicenter PM studies, we plan to have the Trans-Duodenal Barrier ready for the market in 2027.

