May 2026
TDC launches pivotal clinical trial for Trans-Duodenal Barrier
Novel endoscopic treatment method for diabetes and obesity from Mannheim being tested nationwide
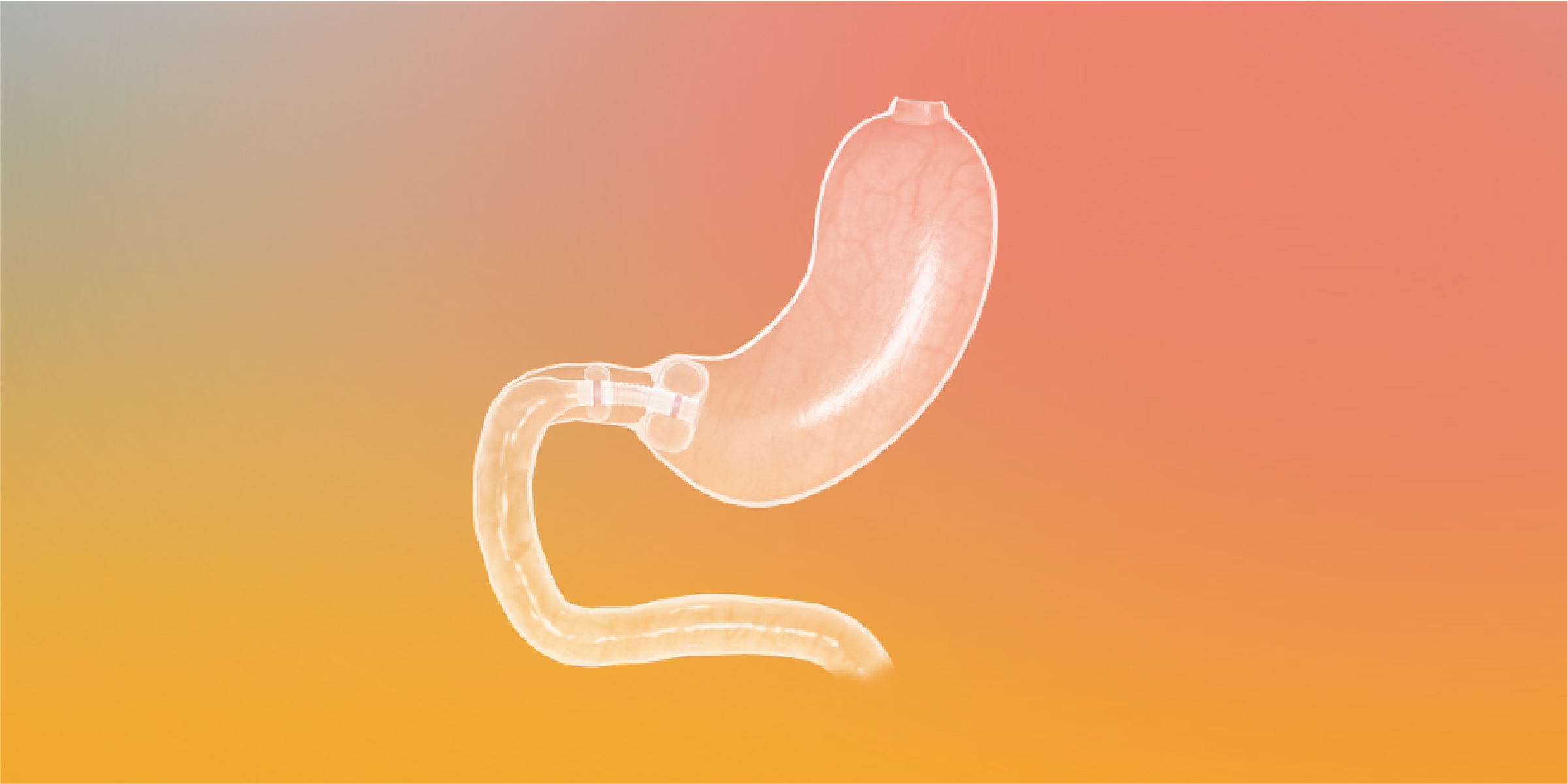
Trans-Duodenal Concepts GmbH started the clinical approval study for its endoscopic implant Trans-Duodenal Barrier on May 4, 2026 following approval by the BfArM and the approval of all responsible ethics committees. The university hospitals in Berlin (Charité), Bochum, Heidelberg-Mannheim, Leipzig, Würzburg and the hospitals in Bamberg, Dresden and Leipzig (St. Georg) are participating in the study with 125 patients under the direction of Prof. Christoph Reissfelder, Mannheim.
It is intended to demonstrate the effectiveness of the implant in terms of improving diabetes metabolism and weight loss, as well as its safety and tolerability.
The study is being organized by novineon CRO GmbH Tübingen and supported by the Federal Ministry of Research, Technology and Space.
Obesity, which affects up to 1 billion people worldwide, is associated with numerous secondary diseases such as liver inflammation, cancer, cardiovascular disease and high blood pressure. Type 2 diabetes mellitus plays a special role in this.
Die TDC unter Leitung von Prof. Georg Kähler ist Entwickler und Hersteller eines endoskopischen Implantates zur nicht-invasiven Therapie von Diabetes und Adipositas. Sie adressiert damit einen der größten medizinischen Märkte mit wachsender soziökonomischer Bedeutung.
The principle of an atraumatic reversible endoscopic bypass represents a global innovation and is also attracting great interest worldwide due to its repeatability and combinability with other forms of therapy.
Contact
Prof. Dr. Georg Kähler
Trans-Duodenal Concepts GmbH
Tannhäuserring 93
68199 Mannheim
Phone +49 179 734 77 34
georg.kaehler@transduodenal.com
